G-20K™
Flow Electroporation® GMP Processing Assemblies
Flow Electroporation Processing Assemblies for GMP and clinical research applications
ExPERT™ GMP Processing Assemblies provide the scale and quality standards required for clinical research and production in GMP clean-room facilities. All Processing Assemblies are produced using high quality medical-grade materials that are thoroughly tested for function, sterility, endotoxin, and particulates. These products are included in our master file at the US FDA and similar regulatory agencies globally to support IND applications.
G-20K™
- Maximum cell recovery at all volumes
- Bioweld compatible; closed process adaptable
- Designed for high-energy transfection
Configuration: Closed system, PVC bags and tubing with luer locking syringe ports
Cell Number: 2.5×108 – 4×109
Volume Range: 5 – 20 mL
Application: GMP applications and clinical research
Catalog: EG020M1-01
Compatible with GTx™
Brand: MaxCyte
SKU: G-20K™
Categories:
Electroporation Technology, Processing Assemblies
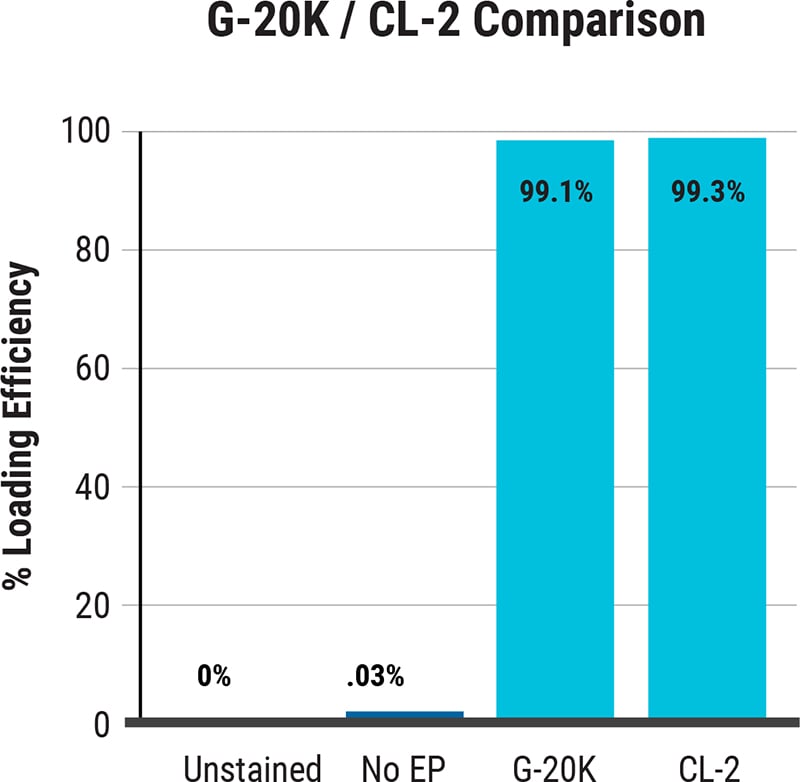
G-20K vs CL-2: K562 cells (1×108 cells/mL) were electroporated using the CL-2 and G-20K processing assemblies to deliver FITC Dextran. The final working volume in each device was 20 mL. Three 100 µL aliquots were taken from each device post electroporation and plated with 3 mL of media for 1 hr. FITC fluorescence was measured using flow cytometry and efficiency determined. All graphs are an average of 3 data point readouts.
Got a question?
Reach out for quick assistance with your needs.

